Please see the accompanying Moderna COVID-19 Vaccine, Bivalent (Original and Omicron BA.4/BA.5) Fact Sheet for Healthcare Providers Administering Vaccine for more information. These expert-vetted articles and infographics provide guidance on the clinical management of COVID-19 patients, ICU administration, procedures, supply chain. 
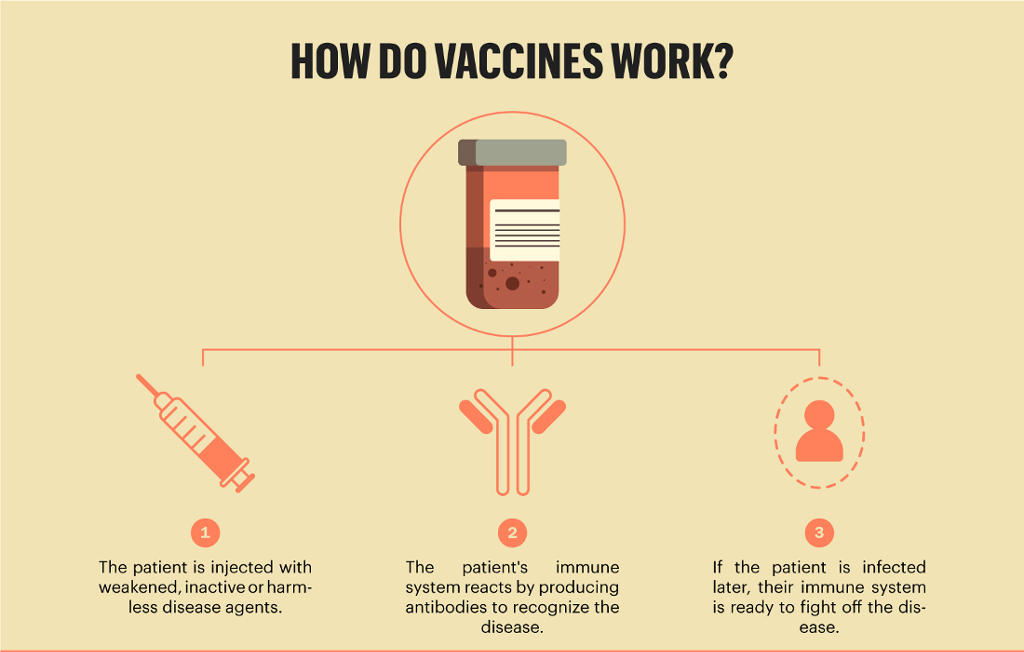
In addition, you can report side effects to ModernaTX, Inc. 24, 2020, and 'the first Moderna shot went into a volunteers arm in Seattle on March 16, 2020,' according. If the information you are looking for is not here, check out our. Moderna delivered the first doses of its Covid-19 vaccine to the NIH for testing on Feb. Find information and resources for each of the available Moderna COVID-19 Vaccines. Reports should include the words “Moderna COVID-19 Vaccine, Bivalent EUA” in the description section of the report. This page answers the most frequently asked questions about COVID-19 vaccine safety. For further assistance with reporting to VAERS, call 1-80. Serious adverse events (irrespective of attribution to vaccination)Ĭases of Multisystem Inflammatory Syndrome (MIS)Ĭases of COVID-19 that result in hospitalization or deathĬomplete and submit reports to VAERS online at. Vaccine administration errors whether or not associated with an adverse event beginning in the fall of 2023, the committee unanimously voted that the vaccine composition be updated to a monovalent. The vaccination provider is responsible for mandatory reporting of the following to the Vaccine Adverse Event Reporting System (VAERS): For the 2023-2024 formulation of the COVID-19 vaccines for use in the U.S. Reporting Adverse Events and Vaccine Administration Errors Limitations of Vaccine Effectiveness: The Moderna COVID-19 Vaccine, Bivalent may not protect all vaccine recipients.Ħ months through 36 months of age: Injection site erythema, pain and swelling axillary (or groin) swelling/tenderness, fever, irritability/crying, loss of appetite and sleepinessģ7 months of age and older: Injection site erythema, pain and swelling arthralgia, axillary (or groin) swelling/tenderness, chills, fatigue, fever, headache, myalgia, nausea/vomiting, and rash Please visit the COVID-19 Public Vaccine Distribution section on the COVID-19 Resource Center for more information. A set of infographics to explain the basics of immune response by the body and the different kinds of vaccines that are being developed around the world to fight against the corona pandemic work. Not all vaccine types are offered at all Northwestern Medicine locations.  
Procedures should be in place to avoid injury from fainting.Īltered Immunocompetence: Immunocompromised persons, including individuals receiving immunosuppressive therapy, may have a diminished response to Moderna COVID-19 Vaccine, Bivalent. Do not administer the vaccines to individuals with a known history of severe allergic reaction (e.g. COVID-19 vaccines are supplied by state and local agencies. Syncope (fainting): May occur in association with administration of injectable vaccines. Published: Preliminary clinical data demonstrated a robust immune response by the companys XBB.1. Source: National Center for Immunization and Respiratory Diseases. The CDC has published considerations related to myocarditis and pericarditis after vaccination, including for vaccination of individuals with a history of myocarditis or pericarditis ( ). Find information and resources for each of the available Moderna COVID-19 Vaccines. Initial vaccine shipments have begun rolling to Virginia hospitals. The observed risk is highest in males 18 through 24 years of age. EUA approval for the Moderna mRNA vaccine is expected soon. Myocarditis and Pericarditis: Postmarketing data demonstrate increased risks of myocarditis and pericarditis, particularly within the first week following vaccination. People ages 5 and older who had all recommended doses of a COVID-19 vaccine can get the Pfizer-BioNTech COVID-19 updated, or bivalent, booster. Monitor vaccine recipients for the occurrence of immediate adverse reactions according to the Centers for Disease Control and Prevention guidelines ( ). Kids ages 6 months through 5 years who got the Moderna COVID-19 vaccine can get an updated, Moderna COVID-19 bivalent vaccine booster. Management of Acute Allergic Reactions: Appropriate medical treatment to manage immediate allergic reactions must be immediately available in the event an acute anaphylactic reaction occurs following administration of the vaccine. J12:33 pm EDT Written by Zacks Equity Research for Zacks -> Moderna MRNA announced that it has submitted a regulatory application to the FDA seeking approval for mRNA-1273.815, its. This webpage will house materials to support COVID-19 Vaccine Providers in successful implementation of the COVID-19 Vaccination Program.Do not administer the vaccine to individuals with a known history of a severe allergic reaction (e.g., anaphylaxis) to any component of the Moderna COVID-19 Vaccine or Moderna COVID-19 Vaccine, Bivalent. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
